- WELCOME TO NEURAGENIX PHARMA
Built on Compliance. Delivered with Confidence
Neuragenix Pharma is a pharmaceutical company purpose-built to secure and manage UK & EU Marketing Authorisations for high-value, essential medicines.
We combine deep regulatory expertise, disciplined execution, and strategic product selection to bring compliant, reliable medicines to market—efficiently and responsibly.
- Wellcome To Healion Pharma
Built on Compliance. Delivered with Confidence
Healion Pharma is a pharmaceutical company purpose-built to secure and manage UK & EU Marketing Authorisations for high-value, essential medicines.
We combine deep regulatory expertise, disciplined execution, and strategic product selection to bring compliant, reliable medicines to market—efficiently and responsibly.

- ABOUT US
Transforming regulatory complexity into commercial opportunity
As a dedicated UK & EU Marketing Authorisation holder, we take ownership of regulatory execution from start to finish. Our expertise spans the full lifecycle of pharmaceutical products—from strategic dossier development to long-term compliance management. We don’t just advise; we execute, ensuring that every submission meets the highest standards of quality and regulatory rigor.
Vision
To become a trusted UK & EU Marketing Authorisation holder and commercial partner for value-driven pharmaceutical products—known for quality, reliability, and execution excellence.
Mission
To secure, protect, and maximise Marketing Authorisations in the UK & EU through scientifically robust, operationally flawless, and strategically aligned with market needs.
Call to questions
+447703184623
CORE CAPABILITIES
Marketing Authorisation Applications
Marketing Authorisation Applications
Regulatory Strategy & Dossier Development
CTD compilation, gap analysis, and scientific justifications built on regulatory intelligence and submission experience.
Lifecycle & Compliance Management
Lifecycle & Compliance Management
In-Licensing & MA Transfers
In-Licensing & MA Transfers
Portfolio & Product Selection
Portfolio & Product Selection
Distribution & Export Enablement
Distribution & Export Enablement
PRODUCT CATEGORIES
Strategic focus across high-value therapeutic areas
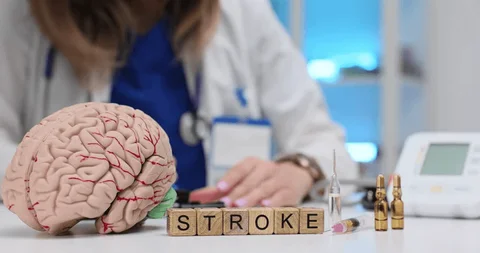
CNS
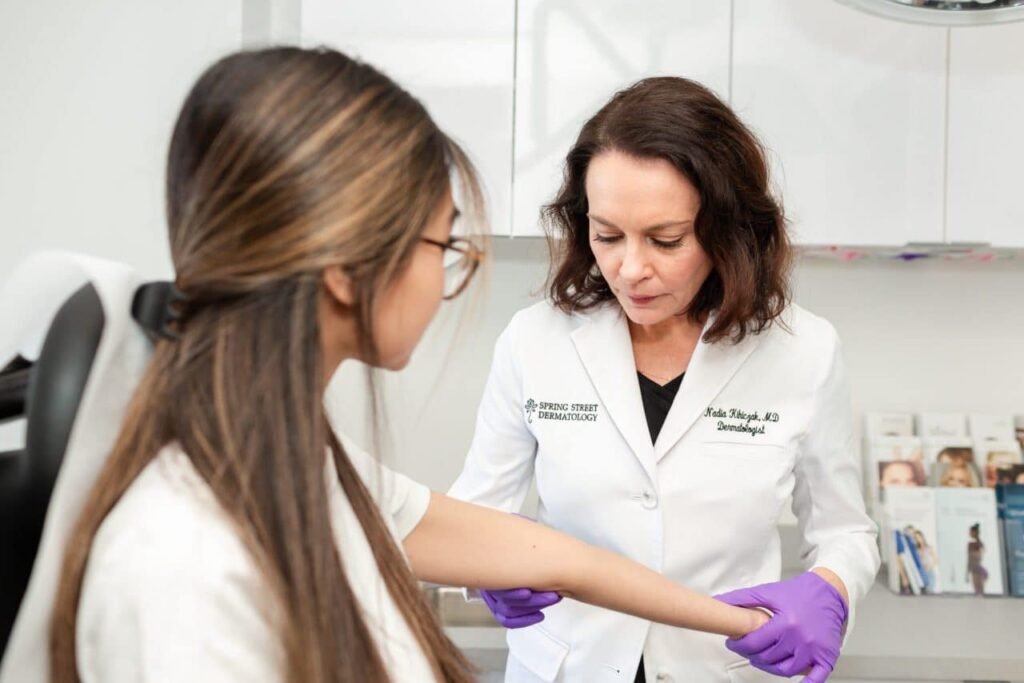
Dermatology

Gastrointestinal

Respiratory

Urology
Anti-Bacterials
CORE CAPABILITIES
Regulatory Ownership, Not Advice
We take responsibility for outcomes—not just recommendations.
Execution Excellence
Structured project management delivering on-time, high-quality submissions.
Value-Driven Strategy
We pursue products that balance public health importance with commercial sustainability.
UK & EU-Focused Expertise
Deep understanding of MHRA expectations, processes, and evolving frameworks.
Compliance by Design
Every process is built to meet regulatory standards today—and tomorrow.
Partnership Approach
Collaborative relationships with manufacturers, distributors, and licensors.
COMMITMENT TO QUALITY
Quality and compliance are not afterthoughts—they are the foundation of everything we do.
Quality Management System
GMP-aligned processes and documentation
Data Integrity
ALCOA+ principles applied throughout
Regulatory Compliance
Continuous monitoring and adaptation
Continuous Improvement
Learning and evolving with the industry
Let's build value together
We partner with CDMOs, CMOs, distributors, product owners, and licensors to bring essential medicines to market through compliant, efficient regulatory pathways.
Let's build value together
We partner with CDMOs, CMOs, distributors, product owners, and licensors to bring essential medicines to market through compliant, efficient regulatory pathways.
